Some see a gearhead. We see the nearly We see the nearly 60 joints performing every tune up
ELOCTATE Prophylaxis Offers Bleed Protection* You Can Count On.

BLEED AND JOINT BLEED PROTECTION* YOU CAN COUNT ON
Preventing bleeds and joint bleeds allows you to get back to all the things you love to do.
(SELECT ONE)
(12 years and older)
(less than 12 years old)
Facts About Bleed Protection
In clinical studies of adults and adolescents using an individualized prophylaxis regimen†:



*ELOCTATE has been proven to help patients prevent bleeding episodes using a prophylaxis regimen.
†In the A-LONG study, 164 adults and adolescents ages 12-65 received ELOCTATE either every 3 to 5 days, once weekly, or on demand.
‡The ASPIRE extension study included 150 people who completed A-LONG. Data from patients treating prophylactically with ELOCTATE for at least 12 months, who had target joints at enrollment in ASPIRE. 234 out of 235 target joints were resolved. A target joint is defined as a major joint with 3 or more bleeding episodes in a consecutive 6-month period. Target joint resolution is defined as 2 or fewer spontaneous bleeds in a 12-month period.
Facts About Bleed Protection
In clinical studies of patients using an individualized prophylaxis regimen†:




*ELOCTATE has been proven to help patients prevent bleeding episodes using a prophylaxis regimen.
†In the Kids A-LONG study, 69 children ages 1-11 received ELOCTATE twice weekly. The ASPIRE extension study included 61 children who completed Kids A-LONG.
‡Data from patients treating prophylactically with ELOCTATE for at least 12 months, who had target joints at enrollment in ASPIRE. 9 out of 9 target joints were resolved. A target joint is defined as a major joint with 3 or more bleeding episodes in a consecutive 6-month period. Target joint resolution is defined as 2 or fewer spontaneous bleeds in a 12-month period.
GET RELIEF. GET CONTROL.
In a clinical study of adults and adolescents treating on demand:


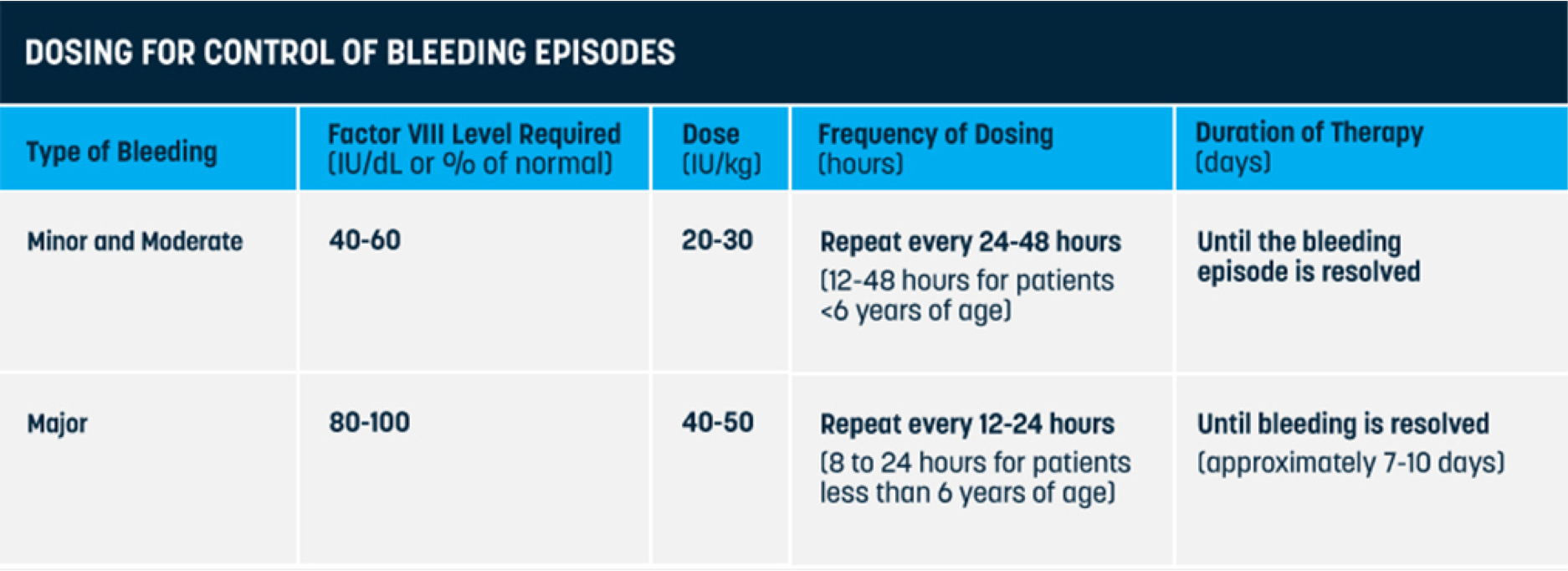
On-demand treatment and control of bleed episodes
+
GET RELIEF. GET CONTROL.
In a clinical study of patients treating on-demand:


On-demand treatment and control of bleed episodes
+
Dosing That Lets You Do You
In clinical trials, 98.8% of patients were able to dose less frequently with ELOCTATE than with their prior standard half-life treatment.
With a variety of vial strength options, ELOCTATE offers individualized dosing and the potential for fewer infusions using a prophylaxis regimen.


See How We Stack Up
In clinical studies, ELOCTATE was shown to last 50% longer in the body than ADVATE®.
ADVATE® [Antihemophilic Factor (Recombinant)] is a registered trademark of Baxalta Incorporated, a Takeda company.

Fc Fusion and You
ELOCTATE is Factor VIII fused with an Fc protein. Fc Fusion utilizes naturally occurring Fc receptors in your body to keep Factor VIII temporarily recirculating in your bloodstream.
CONTACT YOUR CoRe
Sanofi Hemophilia Community Relations and Education (CoRe) Managers offer education to people living with hemophilia and their families. CoRe Managers provide information about living with hemophilia and treatment options. Use our handy CoRe Locator to find the CoRe team member nearest you.
Get informed. Stay informed.
Let's stay in touch. We'll occasionally send you important information on Hemophilia A and ELOCTATE.
LET'S GET STARTEDELOCTATE® [Antihemophilic Factor (Recombinant), Fc Fusion Protein] is an injectable medicine that is used to help control and prevent bleeding in people with Hemophilia A (congenital Factor VIII deficiency). Your healthcare provider may give you ELOCTATE when you have surgery.
IMPORTANT SAFETY INFORMATION AND INDICATION
+
IMPORTANT SAFETY INFORMATION
Do not use ELOCTATE if you have had an allergic reaction to it in the past.
Tell your healthcare provider if you have or have had any medical problems, take any medicines, including prescription and non-prescription medicines, supplements, or herbal medicines, have any allergies, are breastfeeding, are pregnant or planning to become pregnant, or have been told you have inhibitors (antibodies) to Factor VIII.
Allergic reactions may occur with ELOCTATE. Call your healthcare provider or get emergency treatment right away if you have any of the following symptoms: difficulty breathing, chest tightness, swelling of the face, rash, or hives.
Your body can also make antibodies called "inhibitors" against ELOCTATE, which may stop ELOCTATE from working properly.
Additional common side effects of ELOCTATE are headache, rash, joint pain, muscle pain and general discomfort.
If you have risk factors for developing abnormal blood clots in your body, such as an indwelling venous catheter, treatment with Factor VIII may increase this risk.
These are not all the possible side effects of ELOCTATE. Talk to your healthcare provider right away about any side effect that bothers you or that does not go away, or if bleeding is not controlled after using ELOCTATE.
INDICATION
ELOCTATE® [Antihemophilic Factor (Recombinant), Fc Fusion Protein] is an injectable medicine that is used to help control and prevent bleeding in people with Hemophilia A (congenital Factor VIII deficiency). Your healthcare provider may give you ELOCTATE when you have surgery.
PLEASE SEE FULL PRESCRIBING INFORMATIONMANUFACTURED BY
Bioverativ Therapeutics Inc.
Waltham, MA 02451 USA
U.S. License #2078
CLICK HERE TO LEARN MORE ABOUT SANOFI'S COMMITMENT TO FIGHTING COUNTERFEIT DRUGS.
INDICATION
ELOCTATE® [Antihemophilic Factor (Recombinant), Fc Fusion Protein] is an injectable medicine that is used to help control and prevent bleeding in people with Hemophilia A (congenital Factor VIII deficiency). Your healthcare provider may give you ELOCTATE when you have surgery.


![ELOCTATE® (Antihemophilic Factor Recombinant), Fc Fusion Protein]](/Content/images/eloctate-logo.svg)